New Delhi, 1/1(AO Bureau): The Oxford COVID 19 vaccine produced by the Serum Institute of India(SII) has got approval for emergency use by a government-appointed panel. The vaccine will now be sent to the Drugs Controller General of India (DCGI) for further approval.
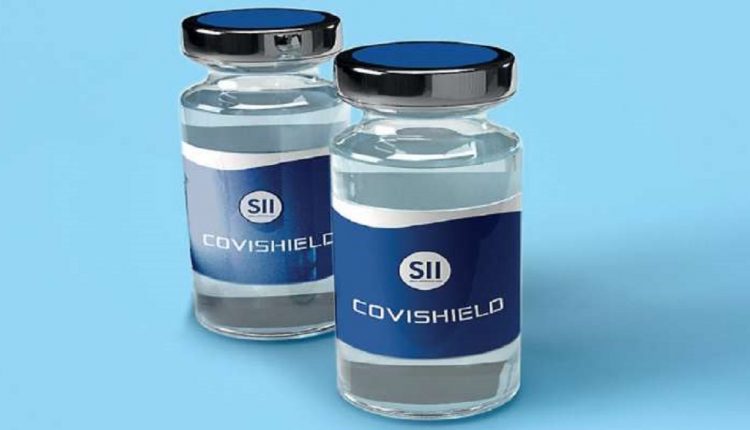
The Serum Institute of India (SII) is making the vaccine Covishield developed by Oxford University and pharma major AstraZeneca. The other vaccine, Covaxin that is developed by Bharat Biotech in association with the Indian Council of Medical Research (ICMR) had also presented its data before the panel. Pfizer had asked for more time to present their data.
The Serum Institute is a partner in Oxford’s vaccine and will sell the vaccine in the country under the name Covishield.
Comments are closed.